What is the Illinois Naloxone Standing Order and how does a patient obtain naloxone from a pharmacy without a prescription? What naloxone dosage forms are available?
Introduction
Every day, more than 130 people in the United States die after overdosing on opioids.1 In response to the growing opioid crisis, Illinois has focused its efforts on promoting the appropriate use and increasing the availability of overdose-reversing drugs, such as naloxone, for individuals with opioid addiction and their families, with the goal of reducing opioid overdose deaths.2
Naloxone (Narcan®, Evzio®) is an opioid-receptor antagonist. It is used to rapidly reverse opiate agonist (i.e. oxycodone, hydrocodone, morphine, fentanyl, codeine and heroin) overdose effects, including respiratory depression, central nervous system depression, and hypotension, by blocking opioid receptors in the brain.2,3,4 Naloxone is safe, effective, and easy to administer, especially with new dosage forms currently available.
In September 2015, Illinois enacted Public Act 99-0480 to expand the public’s access to naloxone. The law authorizes trained pharmacists and first responders to dispense naloxone as a vital component of an opioid overdose intervention plan. As a result, The Illinois Department of Financial and Professional Regulation (IDFPR), the Illinois Department of Public Health (IDPH), and Illinois Department of Human Services (IDHS) issued The Illinois Naloxone Standardized Procedure to provide specific guidance for trained professionals to obtain, dispense, or administer naloxone.5
What is the Illinois Naloxone Standing Order?
The Naloxone Standardized Procedure summarizes how entities may become authorized to obtain, dispense, and administer naloxone hydrochloride for an opioid overdose and includes educational requirements for obtaining the Illinois Naloxone Standing Order.5
The Naloxone Standing Order was created in accordance with the Alcoholism and Other Drug Abuse and Dependency Act (20 ILCS 301/5-23) and Executive Order 17-05.5 It was issued by the Chief Medical Officer of the Illinois Department of Public Health on September 7, 2017 and is renewed annually. The Naloxone Standing Order authorizes trained, licensed pharmacists and overdose education and naloxone distribution (OEND) programs to provide naloxone to individuals who request it to reverse a potential opioid-related overdose without a direct prescription. Opioid Overdose Education and Naloxone programs include law enforcement agencies, drug treatment programs, local health departments, hospitals, urgent care facilities, or other community-based organizations that do not have access to a standing order through their organization.
Pharmacists must complete approved training in order to dispense naloxone as a standing order. Training topics include opioid overdose recognition and prevention, naloxone administration techniques, and the importance of calling 911 after naloxone administration.5 Pharmacists may complete Illinois Department of Public Heath (IDPH) approved training modules (previously approved modules include those developed by Walgreens, CVS, Albertsons, Midwestern University, and IPhA).6 Alternately, applicants may meet requirements by demonstrating an understanding of the Naloxone Standardized Procedures and completing the following training videos:
Pharmacies using the standing order to dispense naloxone must report naloxone-dispensing information to the Illinois Prescription Monitoring Program. In addition, the standing order allows insurers, such as Medicaid and Medicare, to be billed for naloxone, allowing it to be more affordable and thereby decreasing the cost barrier for access to care.
How does my patient obtain naloxone?
You may provide your patient a naloxone prescription that can be filled at any pharmacy or inform them that he/she may obtain naloxone without a prescription at a pharmacy that is authorized to dispense naloxone using a standing order.5 Presently, your patient may contact the pharmacy of their choice to determine if they are enrolled. In the near future, the Illinois Department of Public Heath’s website (http://www.dph.illinois.gov/naloxone) will feature the Illinois Naloxone Map, which will identify the location of participating pharmacies.6
What naloxone dosage forms are available?
| Single-Step Intranasal Naloxone (Narcan®)2 | Intramuscular Naloxone 4 | Auto-Injector Naloxone (Evzio®)3 | |
|---|---|---|---|
| Brand Name | Narcan® Nasal Spray | Evzio ® Auto-Injector | |
| Assembly Required | No | Yes | No |
| Easy to Use | Yes | No | Yes |
| Strength | 4 mg/0.1 ml | 0.4 mg/ml | 2 mg/0.4ml |
| Storage Requirements | Store at 68-77 °F Excursions from 41-104°F | Store at 68-77 °F | Store at 59-77 °F Excursions from 39-104 °F |
| How Supplied | Carton containing two 4 mg/0.1 mL intranasal devices | Single-use 1 mL vials | Carton containing two 2 mg/0.4 mL prefilled auto-injector devices |
| Directions for use | Spray 0.1 mL into one nostril. Repeat with second device into other nostril after 2-3 minutes if no or minimal response. | Inject 1 mL in shoulder or thigh. Repeat after 2-3 minutes if no or minimal response. | Inject into outer thigh as directed by English voice-prompt system. Place black side firmly on outer thigh and depress and hold for 5 seconds. Repeat with second device in 2-3 minutes if no or minimal response. |
How to use Single-Step Intranasal Naloxone (Narcan®)5,7

- Peel back the package to remove the device.
- Hold the device with your thumb on the bottom of the plunger and 2 fingers on the nozzle.
- Place and hold the tip of the nozzle in either nostril until your fingers touch the bottom of the patient’s nose.
- Press the plunger firmly to release the dose into the patient’s nose.
- Repeat if there is no response after 3 minutes.
How to use Auto-Injector Naloxone (Evzio®)5,7

- Pull auto-injector from outer case.
- Pull off red safety guard.
- Place the black end of the auto-injector against the outer thigh, through clothing if needed, press firmly and hold in place for 5 seconds.
- Repeat if there is no response after 3 minutes.
How to use Intramuscular Naloxone 5,7
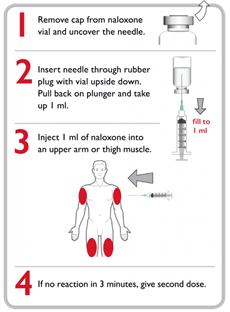
- Uncap the naloxone vial and uncap the muscle needle-syringe.
- Insert the muscle needle through the rubber membrane on the naloxone vial, turn the vial upside down, draw up 1 ml of naloxone liquid, and withdraw the needle.
- Insert the needle into the muscle of the upper arm or thigh of the victim, through clothing if needed, and push on the plunger to inject the naloxone.
- Repeat the injection if there is no response after three minutes.
Conclusion
Naloxone Standing Orders have significantly increased the public’s access to naloxone. To date, 47 states and Washington D.C. have adopted standing orders.8 The National Bureau of Economic Research found that adoption of a naloxone access law was associated with a 9 to 11% reduction in opioid-related deaths.9 Increasing people’s awareness of naloxone availability is essential in expanding its use. To obtain additional information regarding Illinois’ Naloxone Standing Order, see http://www.dph.illinois.gov/naloxone
References
- National Institute on Drug Abuse. Opioid overdose crisis. NIDA. https://www.drugabuse.gov/drugs-abuse/opioids/opioid-overdose-crisis . Published January 22, 2019. Accessed November 21, 2019.
- Narcan [package insert]. Radnor, PA: Adapt Pharma; 2019.
- Evzio [package insert]. Richmond, VA: Kaleo Inc; 2016.
- Naloxone Hydrochloride Injection [package insert]. Eatontown, NJ: West-Ward Pharmaceuticals Corp.; 2018.
- Illinois naloxone standardized procedure. http://www.dph.illinois.gov/sites/default/files/Naloxone-SO-Procedures.pdf . Accessed November 21, 2019.
- Naloxone. http://dph.illinois.gov/sites/default/files/IDPH-Naloxone-FAQ-110117.pdf . Accessed January 30, 2020.
- How to use naloxone and prevent overdose. http://dph.illinois.gov/sites/default/files/images/Naloxone-Brochure-09052017.pdf . Accessed November 21, 2019.
- State Naloxone Access Rules and Resources. SAFE Project. https://www.safeproject.us/naloxone-awareness-project/state-rules/. Accessed February 4, 2020.
- Rees, Daniel, J J, et al. With a Little Help from My Friends: The Effects of Naloxone Access and Good Samaritan Laws on Opioid-Related Deaths. NBER. https://www.nber.org/papers/w23171. Published February 16, 2017. Accessed February 4, 2020.
Kathy Kasiurak, PharmD
Clinical Instructor
University of Illinois at Chicago
College of Pharmacy
The information presented is current as of February 2020. This information is intended as an educational piece and should not be used as the sole source for clinical decision-making.
Posted on Feb. 5, 2020
Last updated on Feb. 6, 2020
Category: Opioids